 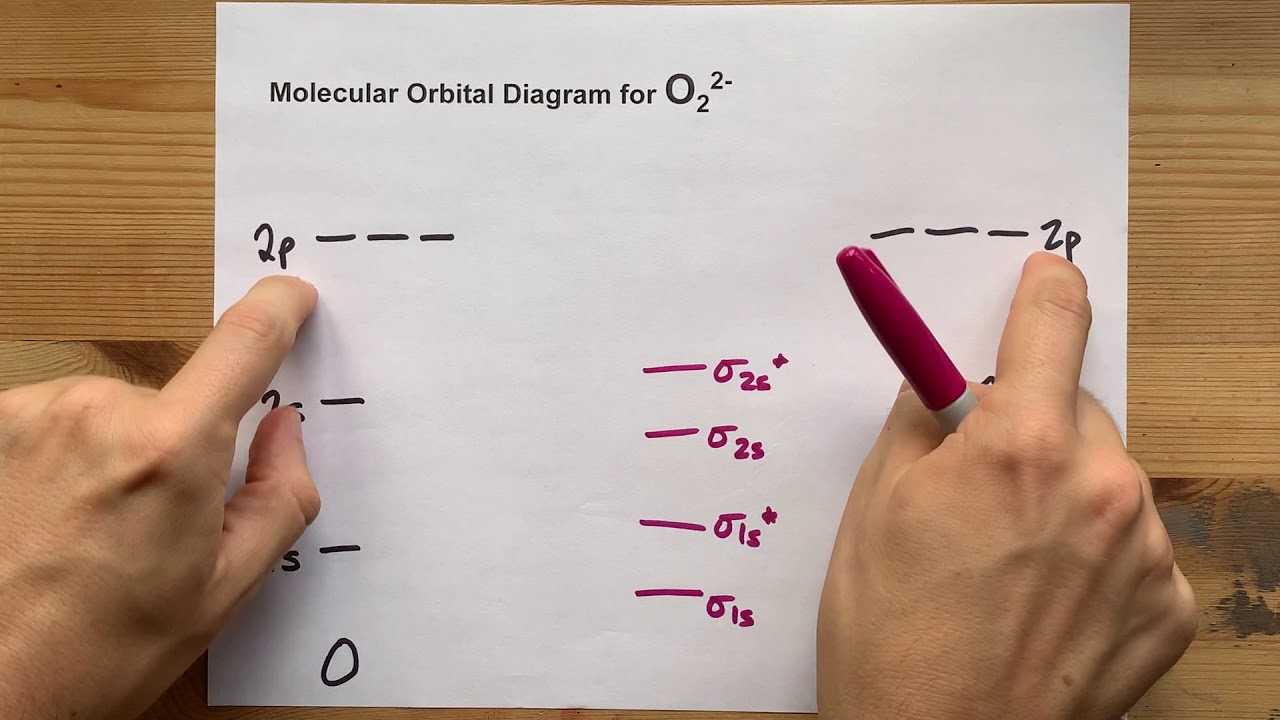
(a) CN (b) CO (c) BeB (d) BC+ Figure 10.47 Molecular orbital diagram for nitric oxide (NO). Write the molecular orbital electron configuration of each, indicating the bond order and the number of unpaired electrons. Identify which combinations of atomic orbitals. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as well as with other compounds. Draw the electron correlation diagram for O2. Oxygen Oxygen is the chemical element with the symbol O and atomic number 8. Please use it to predict the bond order for B2, B2+ ,B2 -, C2, C2+, C2 -, N2. Describe the bonding in the O3 molecule and the NO2- ion using the localized electron model. Solution Verified by Toppr As it can be seen from the MOT of O 2, The electrons in the highest occupied molecular orbital are unpaired therefore it is paramagnetic in nature. Describe the electron dot diagram for a sodium atom. These three electronic states differ only in the spin and the occupancy of oxygen's two antibonding g -orbitals, which are degenerate (equal in energy). Each non-bonding pair is distributed over both oxygen atoms at once in molecular orbital theory, while in Lewis theory each lone pair is isolated to one atom or to localized bonds attached to that atom. The molecular orbital diagram of NO shown in Figure 10.47 also applies to the following species. Solution for Below are the molecular orbital diagrams for B2, C2, and N2. Draw the partial (valence-level) orbital diagram and write the symbol, group number, and period number of the following element. Molecular orbital theory predicts the electronic ground state denoted by the molecular term symbol 3 g, and two low-lying excited singlet states with term symbols 1 g and 1 + g. Create a molecular orbital diagram for the B2+ ion and O22+ ion. from publication: SpinRelated Electron Transfer and Orbital Interactions in. Be sure your diagram contains all of the electrons in the ion, including any core electrons. Download scientific diagram Molecular orbital diagram of O2, H2O, and OH. Still, notice that each orbital is spread across both oxygen atoms at once, and again we see that each non-bonding electron pair in the HOMO is very different in molecular orbital theory compared to Lewis theory. Draw the molecular orbital (MO) electron diagram for the B e 2 + 2 molecular ion. We again fill the orbitals according to Hund’s rule and the Pauli principle, beginning with the orbital that is lowest in energy. \( \newcommand\): Hand-drawn molecular orbital diagram for dioxygen.\( \newcommand\) molecular orbitals, which are truly non-bonding and mostly oxygen in character. To obtain the molecular orbital energy-level diagram for O 2, we need to place 12 valence electrons (6 from each O atom) in the energy-level diagram shown in part (b) in Figure 9.8. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
